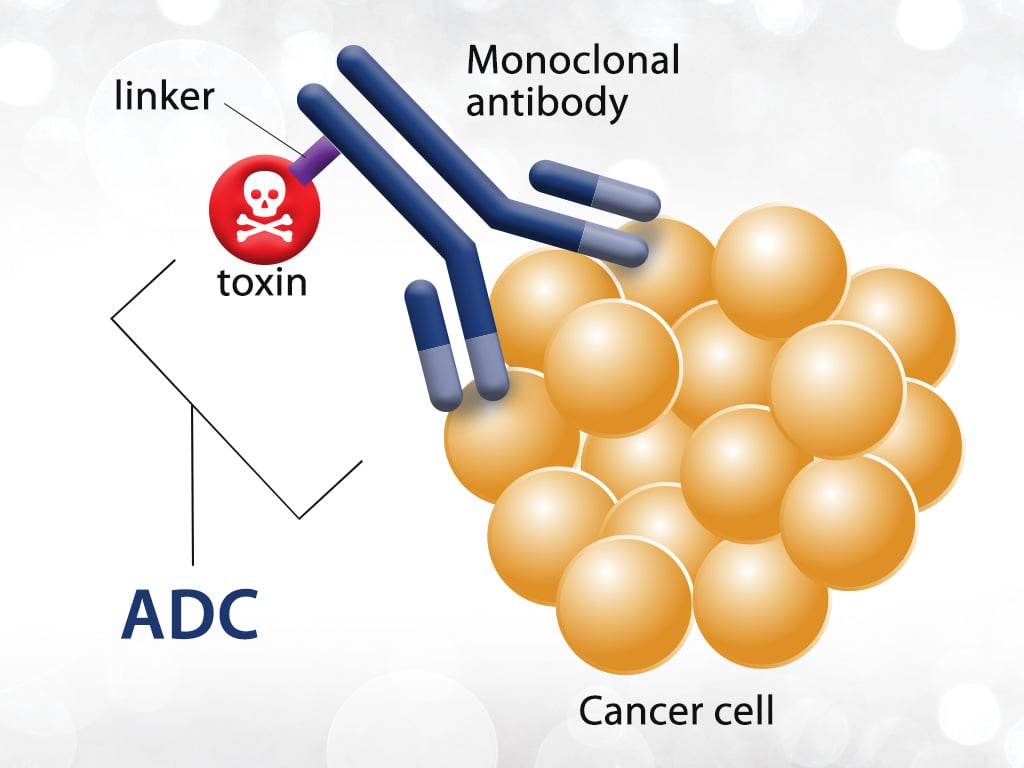
Antibody-drug conjugates (ADCs) are a class of targeted biological agents that couple target-specific monoclonal antibodies with highly lethal cytotoxic drugs through specific linkers, using monoclonal antibodies as carriers to transport small molecule cytotoxic drugs to target tumor cells in a targeted and efficient manner. Therefore, ADC drugs have been one of the popular research directions in the field of tumor precision therapy in recent years. Up to now, 15 ADC drugs have been approved for clinical use worldwide.
The adverse reactions of different ADC drugs differ due to different antibodies and cytotoxic drugs. The related adverse reactions are classified according to the organs and tissues affected as: hematological adverse reactions, infusion-related reactions, neurotoxicity, hepatotoxicity, pulmonary toxicity, gastrointestinal disorders, cardiotoxicity, infections, skin and subcutaneous tissue disorders, tumor lysis syndrome, metabolic toxicity, ocular disorders, and central toxicity.
1. Hematological adverse reactions
Hematological adverse reactions are common with ADC drugs, including complete blood cytopenia, neutropenia, thrombocytopenia, etc. Serious hematological adverse reactions can further increase the risk of bleeding and infection.
A complete blood count check should be performed before the application of ADC drugs. Patients who do not meet the treatment requirements should be administered with caution, and the drugs should be administered only after the blood indicators have returned to normal or after supportive treatment has been given to restore normalcy. Monitor blood counts regularly during treatment and also consider prophylactic medication for secondary prevention.
2. Infusion-related reactions
Infusion reactions are a common adverse reaction in patients treated with ADC medications, with an incidence of 2.5% to 13.0%. The main symptoms include fever, chills, and occasionally nausea, vomiting, pain, headache, dizziness, dyspnea, hypotension, rash, and malaise. Symptoms of severe infusion reactions then include dyspnea, hypotension, croup, bronchospasm, tachycardia, respiratory distress, supraventricular tachyarrhythmias, and urticaria.
Patients at risk should be premedicated with steroidal corticosteroids, acetaminophen, and/or diphenhydramine to minimize the risk of IRR. Monitor for the occurrence of infusion reactions during and for at least 1 h at the end of the infusion. For patients with infusion reactions, the infusion may be promptly interrupted and treated symptomatically with steroid hormones or antihistamines; for patients with severe infusion reactions, permanent discontinuation of the drug is recommended.
3. Peripheral neuropathy
The symptoms of peripheral neuropathy are mainly sensory nerve damage, such as various kinds of hyperalgesia, sensory allergy, sensory inversion and burning pain, and other symptoms of neuralgia, and in severe cases, weakness of limbs, difficulty in squatting, inability to walk, or even bedridden. When patients have peripheral neuropathy, they can be given B vitamins for nerve nutrition treatment; for symptoms of neuralgia, symptomatic treatment such as gabapentin, pregabalin, amitriptyline, venlafaxine or duloxetine can be chosen. If necessary, the neurology department can assist in the diagnosis, differential diagnosis and treatment of peripheral neuropathy.
When more serious peripheral neuropathy (grade 3) occurs during ADC treatment, such as when the patient is unstable due to limb weakness and needs tools to walk, or when the quality of life is still seriously affected by numbness and pain in the limbs after medication for neuralgia, which leads to difficulties in life, ADC treatment should be suspended; if the symptoms improve and the patient is able to take care of himself/herself, consideration can be given to restarting treatment and adjusting the ADC dose to a lower level. If more serious neuropathy (grade 4) occurs, which endangers the patient’s life, ADC treatment should be terminated immediately.
4. Hepatotoxicity
Routinely perform liver function tests and intervene promptly when liver function abnormalities occur.
5. Pulmonary toxicity
While receiving treatment, patients are advised to report cough, dyspnea, fever and/or any new worsening respiratory symptoms immediately, while closely monitoring patients for signs, symptoms and imaging changes of interstitial lung disease, detecting evidence of interstitial lung disease in a timely manner, and requesting respiratory consultation for patients with suspected interstitial lung disease.
- For asymptomatic (grade 1) interstitial lung disease, steroid corticosteroid therapy (e.g., ≥5 mg/kg prednisolone or other hormone of equivalent dose potency) should be considered and may be continued.
- If symptomatic (grade 2 or higher) interstitial lung disease develops, steroid corticosteroid therapy (e.g., ≥1 mg/kg prednisolone or other hormone of equivalent dose potency) needs to be initiated immediately. After stable symptom control and imaging suggestive of improved lung shadow absorption, the steroid corticosteroid dose should be tapered to maintain a relatively long course of therapy (e.g., 4 weeks).
- Patients diagnosed with symptomatic (grade 2 or higher) interstitial lung disease should be permanently discontinued. If symptoms continue to worsen, multidisciplinary consultation and aggressive intervention is recommended to avoid progression to a lethal outcome.
6. Gastrointestinal adverse reactions
Gastrointestinal reactions are common adverse reactions of ADC drugs, including nausea, vomiting and diarrhea, etc. They are usually mild, but if serious GI reactions occur, close attention and active management are required.
7. Cardiotoxicity
Cardiotoxicity is a common toxicity of anti-HER-2 drugs, usually manifested as a decrease in left ventricular ejection fraction (LVEF). Before applying TD and T-DM1, patients should be thoroughly evaluated, including personal and family history, adequately correcting risk factors such as cardiovascular disease, standardizing treatment of combined underlying cardiovascular disease, recording ECG and echocardiogram at baseline, and measuring baseline troponin and natriuretic peptide in patients who have received previous anthracycline therapy.
ECG and cardiac ultrasound should be reviewed dynamically and regularly during treatment, and myocardial markers such as brain natriuretic peptide or amino-terminal brain natriuretic peptide, cardiac troponin I or ultrasensitive troponin should be refined if necessary. Objectively evaluate cardiac function and the risk of associated cardiotoxic events for early detection and timely diagnosis and treatment. For patients with combined hypertension at baseline, angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists and beta-blockers are preferred as antihypertensive agents.
- In patients with asymptomatic cardiac insufficiency, consult a cardiovascular specialist and continue ADC drug therapy and increase the frequency of LVEF monitoring (e.g., once every 4 weeks) on top of angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists and β-blockers.
- If the absolute value of LVEF is <50% (decrease ≥16%), or if it is in the normal range but the decrease in LVEF is ≥10% during treatment, ADC drug therapy should be suspended and angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists and β-blockers should be given, and LVEF should be rechecked in 3-4 weeks, and treatment should be continued after LVEF returns to normal.
- If LVEF decreases irrecoverably or severely, or if symptomatic congestive heart failure occurs, the drug should be permanently discontinued, cardiovascular specialist consultation should be requested if necessary, and heart failure should be promptly diagnosed and treated according to the standard process recommended by the guidelines such as the Chinese Society of Clinical Oncology Guidelines for the Prevention and Treatment of Anthracycline Cardiotoxicity (2020).
Huateng Pharma is dedicated to being your most reliable partner to provide chemical synthesis and high-quality PEG linkers for ADC drugs. We are committed to promoting the progress of your ADC discovery and development projects.