Recently, the FDA has rejected a number of products, one of which is a PDC drug that it has written in the past. In the past, because it was specialized in ADC, the main focus was on the design improvement of the linker. At that time, all the antibodies used were her2 monoclonal antibody. , but if the A on the ADC is replaced with another peptide with tumor-specific recognition, that is, a synthetic cyclic peptide, can another way be found to break through the population limitation of Her2 antibodies? An ignorant idea in my student days, I did not expect to achieve a breakthrough in recent years, but unfortunately suffered a defeat.
On September 23, Oncopeptides announced that the U.S. FDA Oncology Drugs Advisory Committee (ODAC) has completed discussions on the benefit-risk profile of Pepaxto, the first peptide drug conjugate (PDC). “Given the potential harm in overall survival (OS), the failure to demonstrate a benefit in progression-free survival (PFS), and the lack of appropriate dosing, is the benefit-risk profile of Pepaxto favorable for the currently designated patient population?” while the ODAC panel asked The answer was overwhelmingly 14:2 – “No”.
In general, the FDA tends to follow ODAC’s recommendations, which means that Pepaxto’s delisting is a foregone conclusion.
At present, the ADC track has also seen target pushes, overlapping indications, and R&D involution in similar forms. Innovation has become an urgent problem for many manufacturers to solve. Existing companies have taken a different approach and deployed peptide-drug conjugates (PDCs, peptide-drug conjugates) with relatively loose competition.
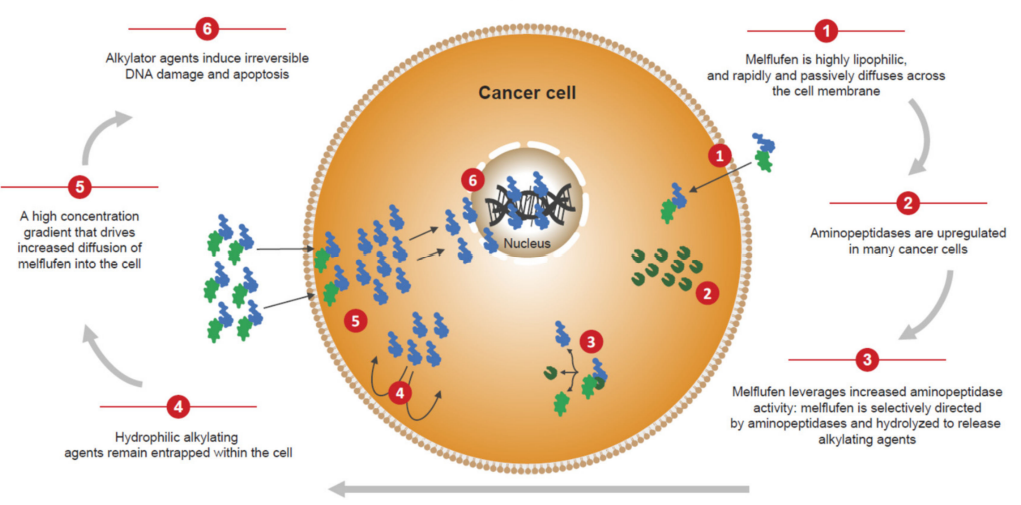
PDC is a novel drug conjugate consisting of a linker, a homing peptide and a cytotoxic payload. The homing peptide can specifically target proteins overexpressed on the surface of tumor cells The receptors thereby deliver cytotoxins to induce tumor cell apoptosis.
Compared with ADC drugs, PDC drugs have the characteristics of small molecular weight, strong tumor penetration, low immunogenicity, large-scale synthesis by solid-phase synthesis, low production cost, and relatively good pharmacokinetics. become a new generation of anticancer drugs.