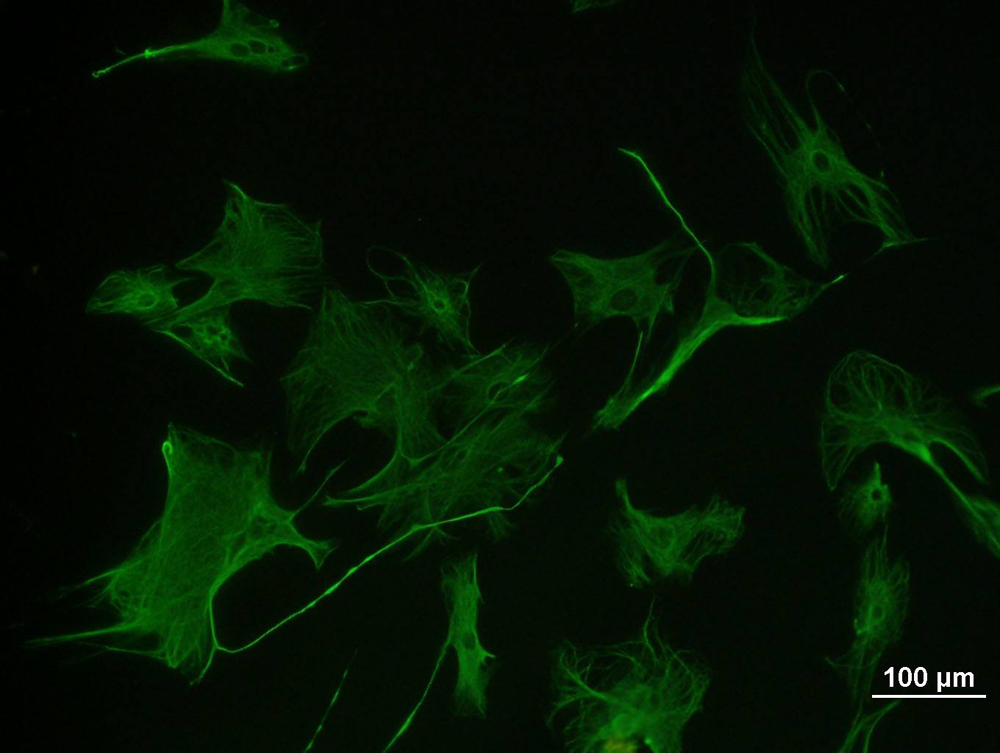
A fluorophore is a fluorescent dye that acts as a fluorescent marker for protein, tissue, and cell labeling for detection by fluorescence microscopy. Fluorophores work by absorbing energy in a specific wavelength region (aka excitation energy range) and then re-emitting that energy in other specific wavelength regions (aka emission energy range).
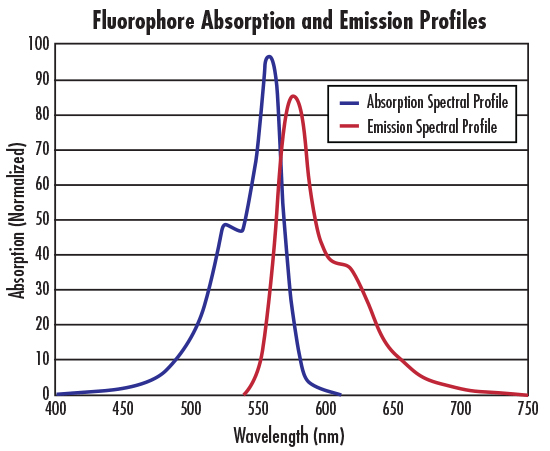
In general, fluorophores are excited or absorb energy by high-frequency illumination (wavelengths in the ultraviolet, violet, or blue spectral regions), and are then excited by lower-frequency illumination (wavelengths in the green, red, or near-infrared spectral regions) ) to emit this energy. Each fluorophore has a wavelength that absorbs energy efficiently, also known as the peak excitation wavelength, λ (nm), and a corresponding wavelength that re-emits the most absorbed energy, also known as the peak emission wavelength, λ (nm) (See Interactive Filter Chart for details). Selecting individual optical filters and wavelengths of maximum transmission will ensure you get the sharpest fluorescence images possible.
In fluorescence microscopy applications, a typical fluorescence microscope setup consists of three filters. See Figure 2 for a typical fluorescence microscope setup.
Filter 1: Excitation Filter
Excitation filters are placed in the illumination path of the fluorescence microscope. Its purpose is to filter all wavelengths in the light source except for the excitation energy range of the fluorophore to be detected. The minimum transmission (%) of the filter is the key to determining the brightness and clarity of the image. For any excitation filter, the minimum recommended transmission is 40%, and the ideal transmission is >85%. The bandwidth of the excitation filter should appear well within the excitation energy range of the fluorophore, and ideally the filter’s center wavelength (CWL) should be as close as possible to the length of the peak excitation wavelength of the fluorophore being detected. Since optical density (OD) is a measure of how well the filter cuts off wavelengths in the bandwidth, the optical density of the excitation filter is the key to the darkness of the image background. The minimum recommended optical density is 3.0, and the ideal optical density is 6.0. Optical filters with optimal center wavelength, minimum transmission (%), and optical density provide the sharpest images, while the filter’s deepest cutoff ensures detection of the weakest emitted signals.
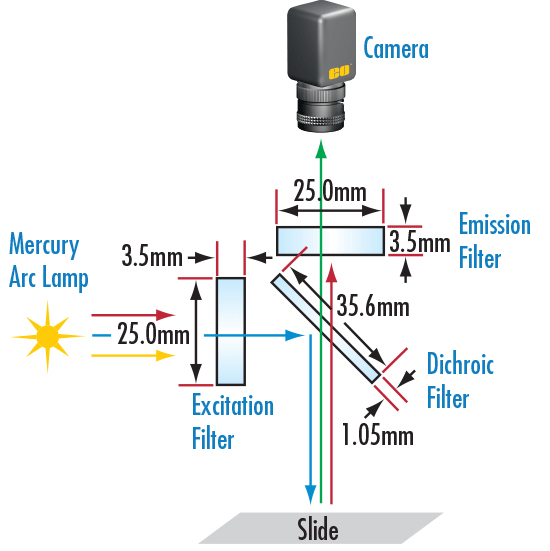
Filter Two: Emission Filter
The emission filter is placed in the imaging path of the fluorescence microscope. Its purpose is to filter the entire excitation energy range of the fluorophore to be detected, as well as transmit the emission energy range of that fluorophore. For emission filters, we recommend the same minimum transmission, bandwidth, optical density, and center wavelength as for excitation filters. Likewise, emission filters with optimal center wavelength, minimum transmission (%), and optical density provide the sharpest images, while the filter’s deepest cutoff ensures detection of the weakest emission signals.
Filter Three: Dichroic Filter or Beamsplitter
A dichroic filter or beam splitter is placed between the excitation filter and the emission filter at a 45° angle. Its purpose is to reflect the excitation signal to the fluorophore to be detected and then transmit the emission signal to the detection device. The most ideal dichroic filter or beamsplitter should be able to transition rapidly between maximum reflectivity and maximum transmission, ideally >95% reflectance for the excitation filter bandwidth and >95% for the emission filter bandwidth. The ideal transmittance is >90%. When selecting filters, the intersecting wavelength (λ) of the fluorophores should be taken into account to ensure fluorescence images with the lowest stray light and the highest signal-to-noise ratio. If you are unsure about choosing the right dichroic filter or beamsplitter, Pingji Optical can assist you in choosing an optical filter that is best for your application.