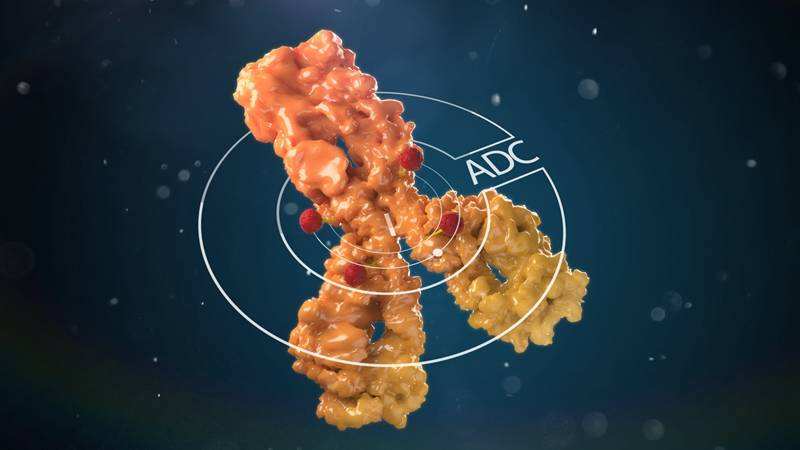
Antibody-drug conjugate (ADC) is to link a biologically active small-molecule drug to a monoclonal antibody through a chemical link, and the monoclonal antibody acts as a carrier to target and transport the small-molecule drug to the target cells.
Introduction to antibody-drug conjugate
Antibody-drug conjugate (ADC) research can be traced back to the 1980s, but it was not until 2000 that the first antibody-drug conjugate (trade name Mylotarg, developed by Pfizer) was approved by the FDA for the treatment of acute myeloid leukemia. Due to the limitations of conjugation technology, targeting, and effectiveness, the complete antibody conjugated drug is unstable in the blood, resulting in lethal toxicity, and was withdrawn from the market in 2010. This casts a shadow over the already unclear ADC drug research.
However, with the improvement of the original technology, Takeda/SeattleGenetics developed a new antibody conjugated drug of brentuximabvedotin (SGN-35, trade name Adcetris,) using its own novel antibody conjugation technology, which was approved by the FDA for use in 2011. Treatment of Hodgkin lymphoma and systemic anaplastic large cell lymphoma. In 2013, the antibody conjugated drug made another breakthrough. Ado-trastuzumabemtansine (T-DM1, trade name Kadcyla) jointly developed by Genentech/ImmunoGen was approved by the FDA for HER2-positive breast cancer, which is the first antibody conjugated drug for solid tumors. . With the successful development of these two drugs, ADC drugs have once again entered the field of research in a fiery state.
ADC drug structure
ADC drugs use specific linkers to link antibodies and small molecule cytotoxic drugs, and its main components include antibodies, linkers and small molecule cytotoxic drugs (SM). Antibody molecules mainly play a role in targeted delivery, and small molecule drugs play an effect. However, some antibodies also have anti-tumor pharmacodynamic effects, such as ado-trastuzumab (ado-trastuzumab) and maytansine (maitansine) in Kadcyla, which have synergistic effects.
Due to the complex structure of ADC drugs, and there are great differences between different ADC drug designs. Even for different drugs of the same target, the differences in their toxicity are obvious due to differences in recognition sites, attachment sites, linkers and small molecules to which they are attached. Therefore, before evaluating the toxicity of an ADC drug, it is necessary to understand the design of the ADC drug.
The ideal ADC drug design needs to be considered
①Antibody selection: clear target, high expression in tumor cells, low expression in normal tissues; support for drug loading, stability, and ability to internalize into cells; good PK properties; less non-specific binding;
②Connection site: generally there are lysine or cysteine residues, which can be modified for directional coupling;
③Linker: stable in circulation and can be released in cells (such as release by enzyme cleavage in lysosome, or release after antibody degradation);
④Cytotoxic drugs: highly pharmacodynamic, non-immunogenic, can be combined with linkers through modification, and the mechanism is clear.