In recent years, chimeric antigen receptor (CAR) T-cell therapy for hematological malignancies has become a major breakthrough in cell therapy. To date, the U.S. Food and Drug Administration (FDA) has approved six CAR-T therapies for the treatment of non-Hodgkin’s lymphoma, B-cell acute lymphoblastic leukemia, and multiple myeloma.
Currently, most clinical trials registered are based on so-called second-generation CARs, which consist of an extracellular antigen-binding domain, a costimulatory domain, and a CD3z signaling domain. Unfortunately, despite the remarkable overall therapeutic effect, there is still a fairly high proportion of patients who do not benefit from CAR-T therapy, the overall response rate is between 50% and 100%, and the subsequent recurrence rate is limited due to the limited durability of the response. up to 66%. In addition, it has been associated with adverse effects such as cytokine release syndrome and neurotoxicity.
Advances in immunology and molecular engineering have promoted the construction of a new generation of CAR-T cells armed with various molecular mechanisms, including additional co-stimulatory domains, safety switches, immune checkpoint regulation, cytokine expression, or therapeutic interference molecules. knockout etc. In this way, the current limitations of CAR-T therapy can be overcome, the efficacy can be enhanced and unnecessary side effects can be reduced. Currently, many clinical trials are currently evaluating the safety and efficacy of novel CAR-T therapies.
The structure of CAR
As CAR-T cell technology becomes more and more mature, the composition of CAR structure, especially the intracellular domain, is also constantly evolving. The first-generation CAR-T cell structure contains a high-affinity single-chain antibody (scFv) to a tumor-associated antigen, a transmembrane domain, and a unique intracellular TCR-ζ or FCR-γ domain. G1 has limited clinical benefit mainly due to its low proliferative capacity and limited cytotoxicity in vivo. To overcome this shortcoming, second-generation CAR constructs are equipped with additional intracellular co-stimulatory domains, most notably CD28 or TNFRSF member 4-1BB. These CARs have proven to be highly effective, and several CAR-T products have received regulatory approval for the treatment of hematological malignancies.
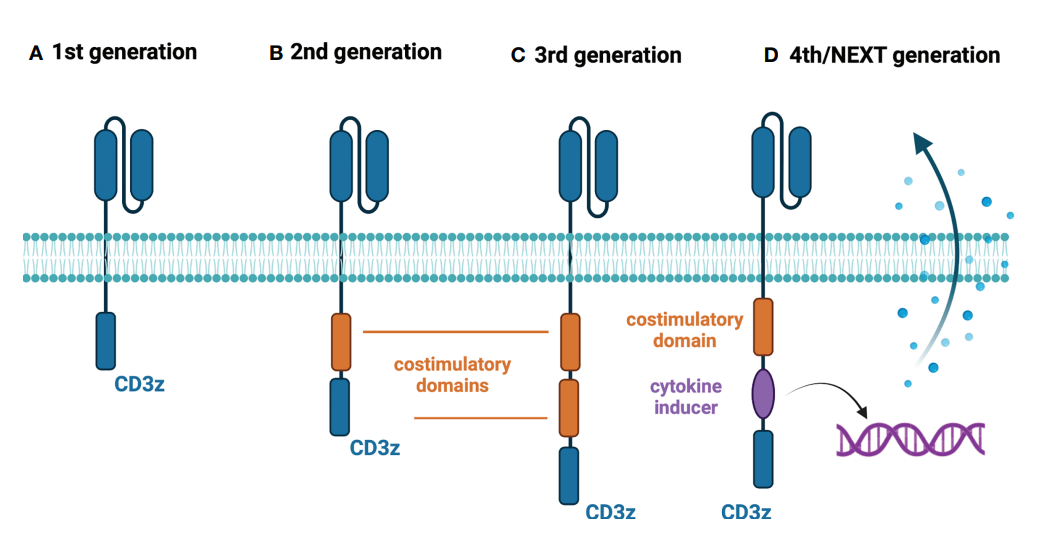
The third-generation CAR-T structure contains a combination of costimulatory domains, while the fourth- and fifth-generation CAR-T cells contain additional costimulatory domains that induce the expression of cytokines (such as IL-12) upon antigen recognition, or contain intracellular domains of certain cytokine receptors (eg, truncated IL-2 receptor beta chain and STAT3-binding portion).
Armed CAR-T cells regulated by immune checkpoints
Immune checkpoint modulation in CAR-T therapy aims to circumvent the suppressive tumor microenvironment. In hematological malignancies, all clinical trials evaluating this approach have explicitly relied on disrupting the programmed cell death protein 1 (PD-1) pathway. Despite the relatively small number of trials (n = 8), individual studies have shown unique ways to modulate PD-1 signaling.
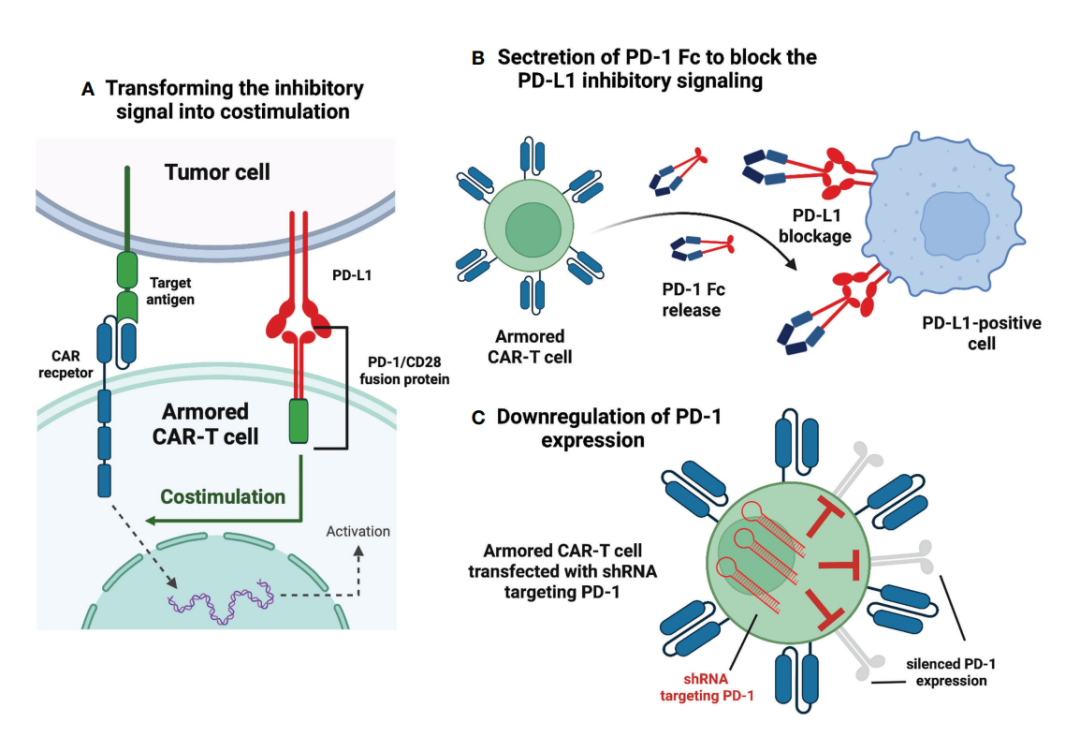
In the NCT03258047 clinical trial, the researchers used an innovative approach in which PD-1 was fused to the intracellular CD28 activation domain. Therefore, the combination of PD-L1 and PD-1 is converted into an activation signal, which is expected to produce a more effective anti-tumor effect. The study involved 17 subjects with B-cell non-Hodgkin’s lymphoma. The results showed that the complete response rate (CR) and objective response rate (ORR) were 41.2% and 58.8%, respectively. Compared with the approved second-generation CAR-T therapy for relapsed/refractory (r/r) DLBCL (CR 52%, ORR 72%), no advantages were shown. Additionally, the NCT03932955 clinical trial is investigating the same approach to immune checkpoint modulation, with no results so far.
Another approach explored to disrupt PD-1 signaling is to reprogram CAR-T cells to secrete PD-1-Fc fusion proteins. Two studies are currently evaluating the safety and efficacy of this approach in r/r multiple myeloma (NCT04162119) and r/r B-cell lymphoma (NCT0416 3302). However, no clinical data have been published so far.
In clinical trial NCT04836507, researchers tested anbalcabtagene autoleucel (Anbal-cel) in patients with r/r large B-cell lymphoma (LBCL). This new CAR-T cell product knocks out PD-1 and TIGIT receptors. Anbal-cel showed impressive results with a CR of 78%. In addition, PD-1 knockdown was also evaluated in the NCT03208556 clinical trial.
In addition to PD-1 pathway regulation, the NCT04037566 clinical trial is evaluating another armed CAR-T cell construction. CAR-T cells with reduced expression of hematopoietic progenitor kinase 1 (HPK1), a negative intracellular immune checkpoint, showed promising preliminary results. Among all enrolled patients, 72.7% achieved CR or CRi, comparable to FDA-approved anti-CD19 therapy.
TRUCKs – CAR-T cells expressing cytokines
T cells redirected for universal cytokine-mediated killing (TRUCKs) are a new generation of CAR-T cells designed to express certain cytokines to enhance the anti-tumor efficacy of CAR-T cells, improve their persistence, and change the characteristics of the tumor microenvironment.
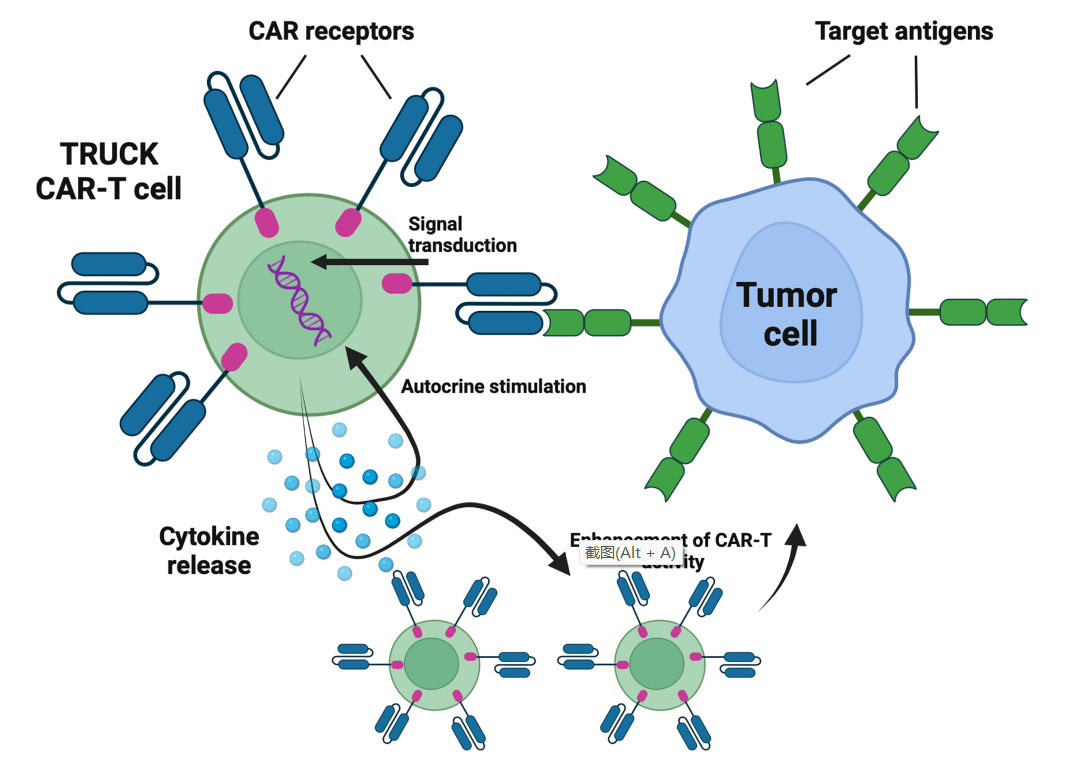
Currently, six clinical trials are evaluating TRUCKs for the treatment of hematological malignancies. Four clinical trials (NCT04381741, NCT03929107, NCT04833504, NCT03778346) are evaluating CAR-T cells expressing IL-7 and the chemokine CCL19. IL-7 promotes the proliferation and survival of T cells, while CCL19 guides the tumor-homing of CAR-T cells. The results showed that the CR rate of patients with r/r DLBCL was 4/7, and the ORR was 5/7. Additionally, NCT03778346 explored the same approach in r/r multiple myeloma (MM) patients, in which two enrolled patients achieved CR (100%).
Another CAR-T cell that secretes the cytokine IL-18 is being evaluated in the NCT04684563 clinical trial. IL-18 is expressed in CAR-T cells to enhance CAR-T cell proliferation and anti-tumor activity. However, IL-18 is also associated with tumor progression, so long-term results regarding the safety of IL-18-expressing CAR-T cells in the clinic remain to be seen.
The NCT03602157 clinical trial represents a different approach to exploit cytokine signaling in CAR-T cells. The researchers constructed an anti-CD30 CAR-T cell expressing CCR4 for the treatment of r/r Hodgkin’s lymphoma and cutaneous T-cell lymphoma (CTCL). The receptor binds to CCL17, thereby improving the transport of CAR-T cells to tumor sites. Preliminary results from the Hodgkin lymphoma cohort trial were promising, with 6 enrolled patients achieving CR (75%) and all patients achieving ORR (100%). Unfortunately, no one in the CTCL group achieved remission.
Safety switch CAR-T cells
The treatment-related toxicity associated with conventional CAR-T therapy has promoted the construction of safety switch CAR-T cells. The new technique enables researchers to incorporate a safety switch to eliminate CAR-T cells by inducing apoptosis, complement-dependent cytotoxicity (CDC), or antibody-dependent cellular cytotoxicity (ADCC) after exogenous drug administration.
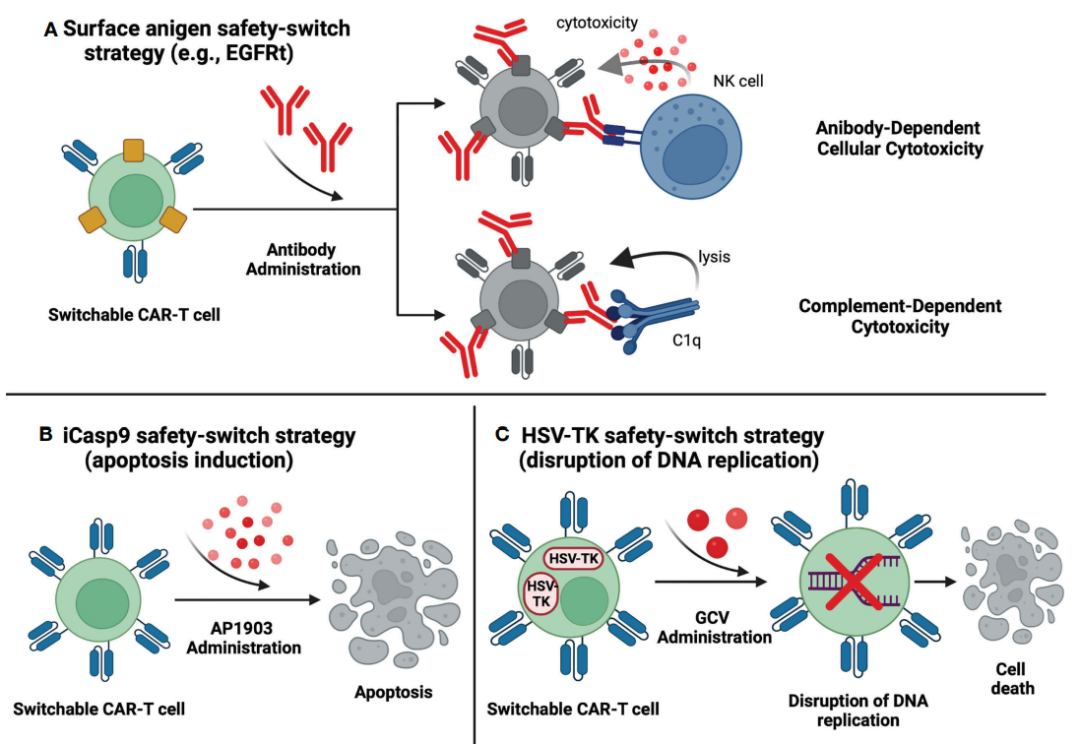
Incorporating truncated epidermal growth factor receptor (EGFRt) into CAR-T cells is a common safety switch approach in clinical trials. EGFRt is targeted by cetuximab, which can be removed by CDC or ADCC. As of August 2022, 20 clinical trials are investigating EGFRt-based CAR-T cells in various hematological malignancies. Currently, there are no available data on the mechanisms that initiate the EGFRt safety switch in humans. The lack of such reporting may be due to the absence of life-threatening adverse events during the investigation period.
In terms of drug efficacy, in CD19-positive malignant tumors, clinical trials have shown different preliminary results. The NCT03085173 study reported an overall CR rate of 57%, with a CR rate of 88% in DLBCL patients and 22% in CLL patients. In clinical trials on patients with B-NHL, the complete response rate was 42% in NCT02706405, 45% in NCT02153580, and 75% in NCT01815749.
The RQR8 suicide gene is a similar approach to control CAR-T cells after treatment administration. The RQR8 gene encodes a cell surface protein derived from CD20 and CD34 epitopes. This strategy can eliminate CAR-T cells through CDC or ADCC after administration of rituximab. Currently, the NCT03590574 clinical trial is studying RQR8-based CAR-T cells in peripheral T-cell lymphoma (PTCL). Preliminary results showed an ORR of 67%, with 56% of patients achieving a complete metabolic response (CMR). Another trial, NCT03287804, was terminated due to unsatisfactory initial results in multiple myeloma.
The addition of inducible caspase 9 (iCasp9) is a unique method of CAR-T safety switch. After administration of AP1903, specifically modified caspase 9 dimerizes and triggers the apoptotic pathway. Currently, 17 clinical trials are evaluating iCasp9-based CAR-T cells, but only 4 of them have provided initial data. The NCT03016377 clinical trial reported neurotoxicity in a patient with ALL after CAR-T infusion. After AP1903 administration, the symptoms were completely relieved, and the only adverse event was a grade 2 bilirubin increase that lasted for three days. Interestingly, despite eliminating more than 90% of CAR-T cells, the researchers observed a clinically significant anti-leukemia response. In the NCT02274584 trial, only one published case report of a patient with Hodgkin’s lymphoma showed a temporary partial response. In addition, the NCT03125577 trial reported data on 4 patients, all of whom experienced CR after CAR-T infusion.
Another suicide gene that can be used as a safety switch for CAR-T therapy is the herpes simplex virus thymidine kinase (HSV-TK) Mut 2 gene, the product of which is targeted by the prodrug ganciclovir (GCV). HSV-TK converts GCV to GCV triphosphate, leading to cell death by disrupting the replication process. HSV-TK-based CAR-T cells were evaluated in the discontinued NCT04097301 trial. Ultimately, only two patients were enrolled and none responded to treatment. The safety switch was not activated due to lack of T cell-associated toxicity.
Universal CAR-T cells
Today, conventional CAR-T products are manufactured from autologous T cells from patients eligible for treatment. This approach has several limitations, including long manufacturing times, difficulty mobilizing appropriate numbers of T cells, and reduced T cell quality in severely treated patients. However, the generation of allogeneic CAR-T cells by molecular engineering can overcome the above obstacles. To construct a universal CAR-T, major histocompatibility complex (MHC) and T cell receptor (TCR) molecules need to be removed from donor-derived cells.
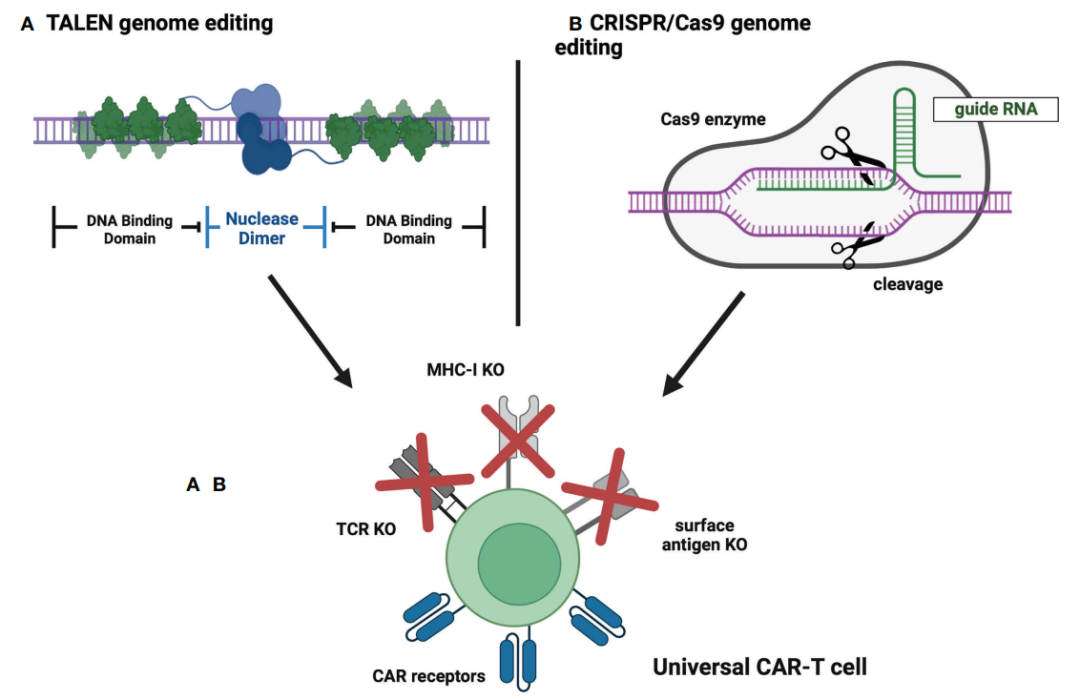
The preliminary safety and feasibility of CRISPR/Cas9 technology in T cell engineering have been demonstrated in the first-in-human trial NCT03399448. As of August 2022, in ClinicalTrials. Five clinical trials evaluating anti-cannibalistic CAR-T cells have been registered on gov. In NCT04502446, CAR-T cells targeting the CD70 antigen were designed to eliminate the expression of TCR, MHC-I and CD70 to eliminate cannibalism and improve efficacy. The results showed that the ORR was 47%, while the CR was 20%, and no serious adverse events were observed. In addition, the NCT04264078 trial provided data on the use of universal CAR-T cells in patients with r/r T-ALL. Disruption of TCR and CD7 genes by CRISPR/Cas9 to prevent GvHD and cannibalism. RESULTS: CR was 80% (4/5), but all subjects were treated with severe CRS (grade 3 in 4 patients and grade 4 in 1 patient). The results of subsequent enrolled patients confirmed the high efficacy of the treatment, with a CR of 83% (5/6), while the safety results were consistent with previous observations.
summary
With the accumulation of research data and the wide application of molecular engineering, the new generation of CAR-T cell therapy is gradually becoming mature in clinical practice. The most successful next-generation CAR-T cells are likely to be universal allogeneic CAR-T cells (manufactured using CRISPR/Cas9 technology), characterized by immune checkpoint resistance and expressing cytokines that transport T cells to tumor sites . Additionally, such structures would contain safety switch mechanisms to manage potential toxicity. This combination may overcome the current limitations of CAR-T cell therapy and help improve patient outcomes.
references:
1. Nextgenerations of CAR-T cells-new therapeutic opportunities in hematology? Front Immunol. 2022 Oct 28;13:1034707.








