With the rapid growth of bioinformatics knowledge related to peptides and proteins, peptide-based drug design strategies have become increasingly active recently. In the past, peptide-based drugs have not been widely used due to serious problems such as low selectivity and rapid degradation of peptides in biological systems. However, a variety of interesting peptide-based therapies have recently emerged, showing excellent antitumor effects. Various newly developed peptide-based drugs are expected to be a promising new class of antitumor therapeutics.
Apoptosis-related peptide drugs
Several polypeptides have been reported to induce apoptosis in cancer cells. For example, an anticancer peptide called Ra-V (deoxybouvardin) can induce mitochondrial apoptosis in tumor cells, followed by loss of mitochondrial function. Another pentapeptide, Dolastatin 10, was able to induce apoptosis by upregulating cytochrome c and Bax.
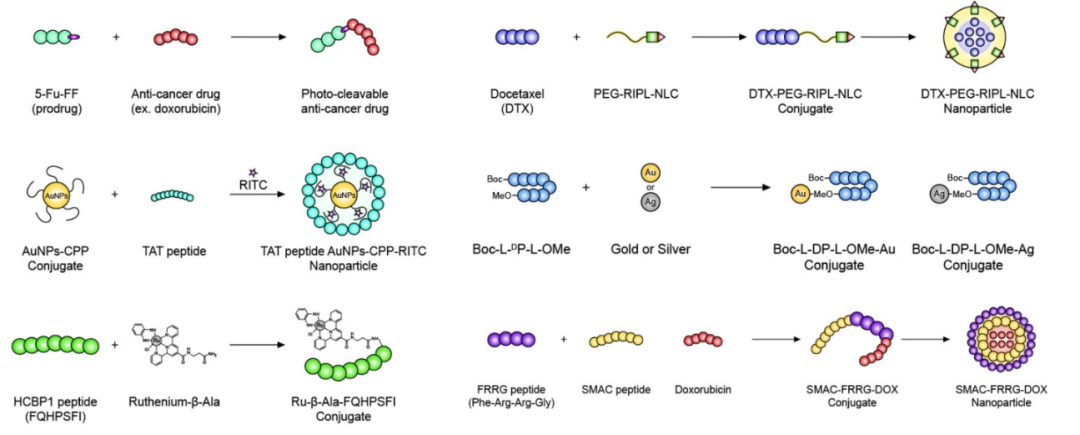
KLA (KLAKLAKLAK) is an interesting pro-apoptotic polypeptide that induces programmed cell death by disrupting the mitochondrial membrane. Bioconjugates containing KLA polypeptides can be applied to the development of various anticancer therapeutic drugs. For example, KLA can be combined with the tumor-homing polypeptide IRGD (CRGDKGPDC) to increase the permeability of tumor tissues and cells. The potent antitumor efficacy of recombinant KLA-IRGD conjugates with high tumor selectivity and low systemic toxicity has been demonstrated in mouse models (Figure 1). These studies may lead to the development of novel targeted PDCs.
Another example of KLA-based therapy is penetratin (a cell-penetrating polypeptide)-KLA conjugate. To overcome the low cellular permeability of KLA polypeptides, penetratin is linked to KLA polypeptides via disulfide bonds, resulting in high cellular permeability and cytotoxicity, even at low concentrations; and penetratin-KLA conjugates have normal cellular mitochondrial effects It is not big, showing its therapeutic potential as a novel polypeptide drug conjugate.
Peptides for tumor enrichment
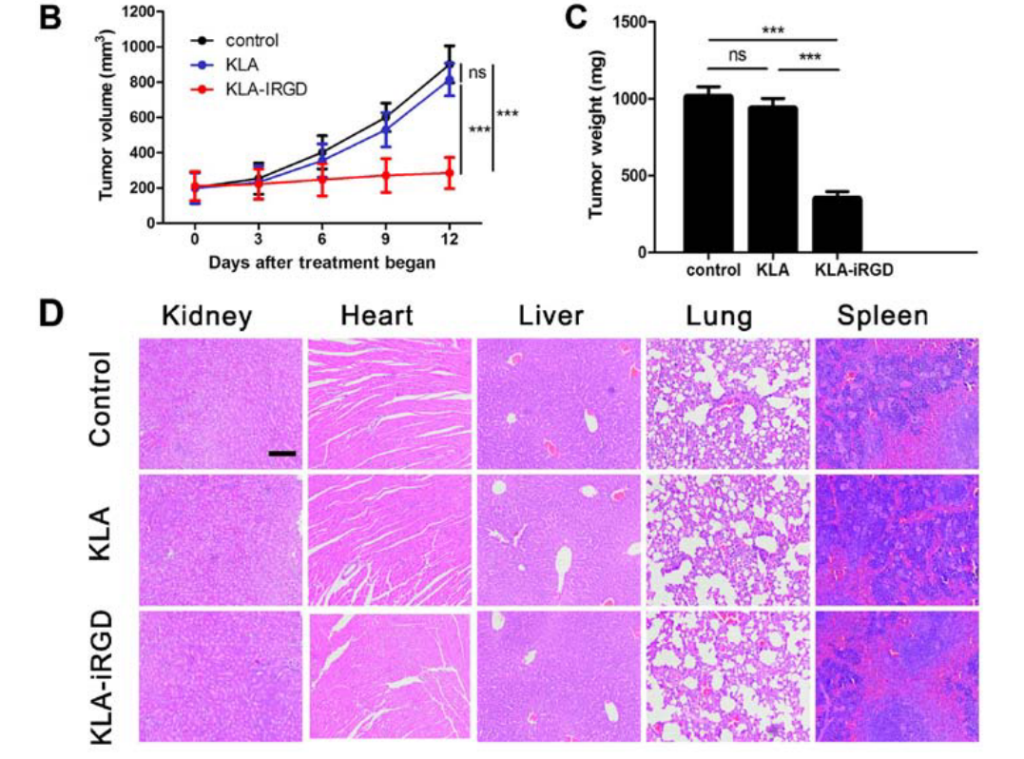
A number of recent studies have introduced various methods for targeting tumors with peptides. For example, the results of Redko et al. showed that S-S bridged cyclic peptide (ALOS-4) is specific for the integrin avβ3, which is highly expressed in human metastatic melanoma. In this study, ALOS-4 was coupled to the topoisomerase I inhibitor camptothecin. The results show that the conjugate has potent antitumor activity in human metastatic melanoma cells, while being less toxic to normal cells. In addition, Brunetti et al. conjugated the four-branched polypeptide NT4 to paclitaxel (PTX). NT4 polypeptides can selectively bind to tumor membrane sulfated glycosaminoglycans. It is highly selective for cancer cells when combined with paclitaxel, a combination that allows it to exhibit more potent antitumor activity than paclitaxel alone (Figure 2).
Albumin is an interesting target for tumor targeting and chemotherapeutic drug delivery. It is an endogenous protein that circulates in the blood longer than other plasma proteins. Therefore, it is not toxic or immunogenic in the body and can be taken up by rapidly growing tumor cells as a source of amino acids and energy. Yousefour et al. coupled a G protein-derived albumin-binding domain to doxorubicin (DOX) via a pH-sensitive linker. This study showed that the ABD-DOX conjugate had a longer half-life in plasma than DOX alone and accumulated four times the amount of DOX in tumors. In another study, the tumor-targeting effect of albumin-binding peptides was investigated. In in vitro studies, the bioconjugation based on albumin-binding peptide (DICLPRWGCLW) to form stable albumin complexes can play a tumor targeting role, and also showed high tumor targeting in the SCC7 tumor-bearing mouse model, The half-life was also significantly increased. Based on the positive correlation between albumin uptake and tumor growth, albumin conjugates are expected to be a novel therapeutic strategy targeting tumor tissues.
Peptide Drugs for Cancer Immunotherapy
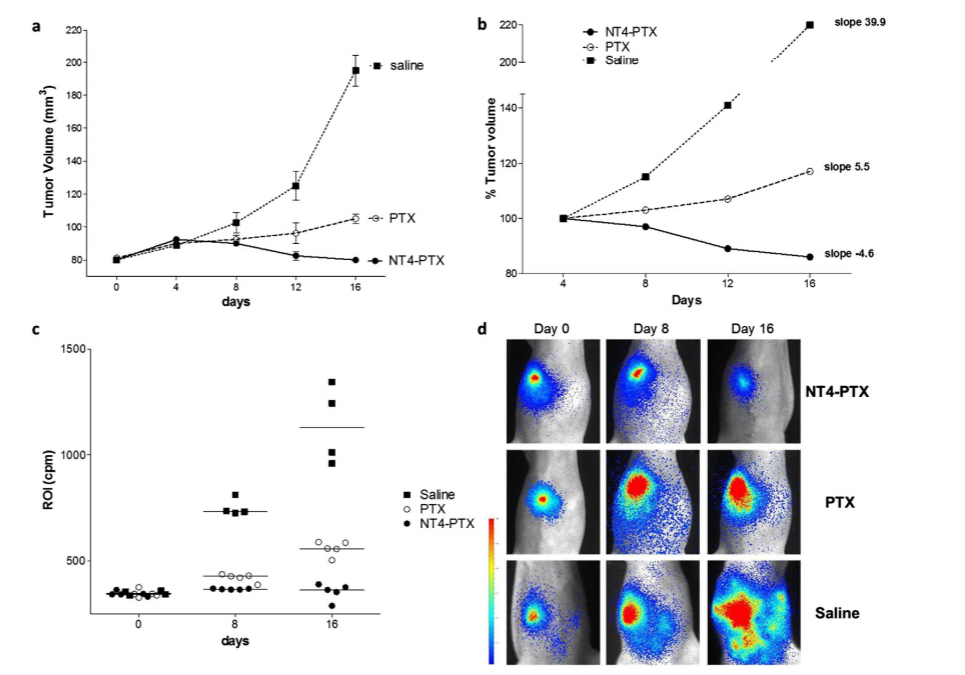
The treatment of cancer by controlling the function of immune cells is also a research hotspot of peptide drugs. PD-L1 is often overexpressed on a variety of cancer cells, and binds to PD-1 expressed on activated T cells, enabling cancer cells to escape from immunity. Inhibition of PD-1/PD-L1 interaction has become a research hotspot in tumor immunotherapy. Among immune-related peptides, D-PPA [NYSKPTDRQYHF] is a PD-L1-binding peptide developed by Chang et al. Studies have shown that hydrophilic D-type polypeptide (D-PPA) itself is not cytotoxic; however, by blocking the PD-1/PD-L1 interaction, it inhibits tumor growth and prolongs animal survival in CT26 tumor-bearing mice (Figure 3).
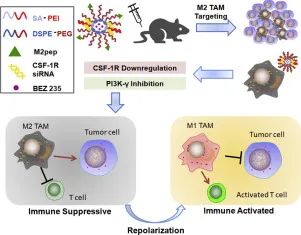
Macrophages are immune cells that play an important role in innate and adaptive immunity. The CSF-1/CSF-1R pathway is involved in the survival and differentiation of macrophages. In a recent study, a polypeptide named M2pep[Cys-YEQDPWGVKWWY] was modified to improve specificity against M2 TAMs. Li et al. demonstrated that co-encapsulation with CSF-1R siRNA and M2pep could target M2 TAMs to remodel to the M1 phenotype, thereby increasing antitumor immune responses. This antitumor immune response effect was confirmed in an animal model of pancreatic cancer (Figure 4).
Peptide drugs conjugated with small molecule cytotoxic drugs
Under the concept of drug conjugates, the combination of polypeptides with cytotoxic drugs or other therapeutic drugs can be used to develop polypeptide-based therapeutics. Typically, cytotoxic drugs that inhibit tumor cell growth or cell function are used in anticancer therapy. Unmodified cytotoxic drugs tend to inhibit rapidly dividing normal cells in the body, not just cancer cells. In various studies, these cytotoxic drugs can be combined with peptides with additional functions. For example, in anticancer therapy, platinum-based cisplatin chemotherapy is one of the most widely used cancer treatment strategies. However, long-term use in patients reduces its efficacy due to cisplatin resistance. The use of peptides may be an important approach to addressing drug resistance-related issues. For example, the findings showed that cisplatin and peptide conjugates induced high accumulation of cisplatin in tumors and metastases-containing organs, and significantly reduced the systemic toxicity of cisplatin.
Currently, some peptide-drug conjugates are undergoing clinical research for the treatment of refractory tumors. On February 27, 2021, the first peptide-conjugated drug melflufen (Melphalan flufenamide, PEPAXTO®) was approved by the FDA for the treatment of multiple myeloma. Since melflufen is highly lipophilic, it is readily taken up by targeted cancer cells. Once internalized, the melflufen molecule can be cleaved by aminopeptidases, which are highly expressed in multiple myeloma cells, and release the alkylating agent-toxic drug, which in turn causes DNA damage and apoptosis in tumor cells.
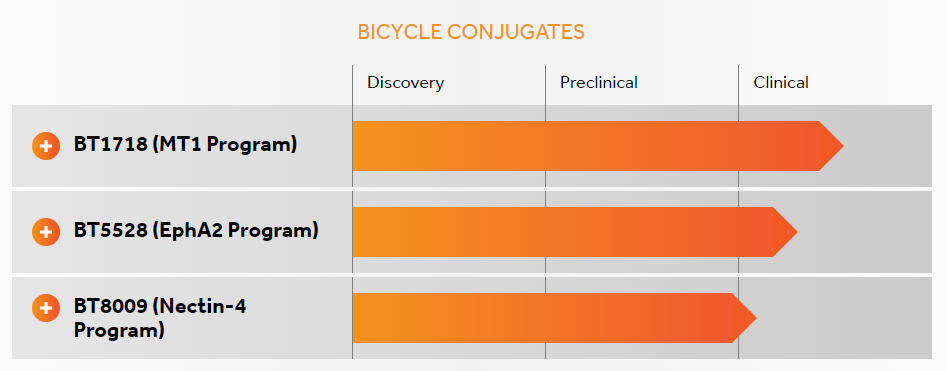
The leader in the layout of PDC at the front end of the industry is Bicycle Therapeutics. The company has uniquely designed the peptides in the peptide-drug conjugates into Bicycle, which is a short peptide with two loops formed by a short linear peptide under the action of a “scaffold”. The molecular weight of Bicycle is 1.5~2KDa. It has the advantages of good tissue permeability and renal clearance, high affinity and selectivity. The large molecular footprint can make the interaction between proteins targeted, which combines the pharmacology of biological drugs. The advantages of pharmacokinetics and the pharmacokinetics of small molecule drugs, without immunogenicity. The company’s PDCs in the form of BTCs (bicycle-toxin conjugates) are currently in multiple clinical trials, including BT1718, BT5528 and BT8009, for the treatment of a range of cancers (Figure 5).
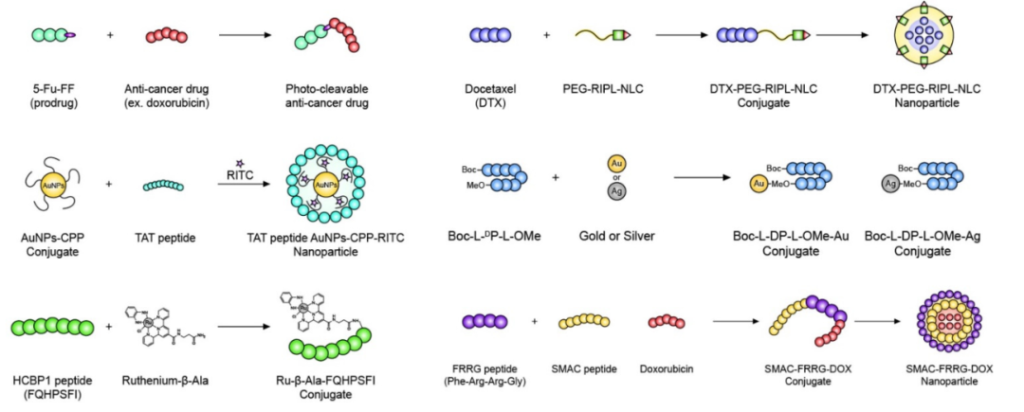
Various other forms of peptide-drug conjugates are also under active development as anticancer drugs (Figure 6). These peptide therapies are expected to outperform most traditional anticancer small molecules, antibodies and ADCs.
Summarize
Due to the advantages of peptides, peptide drugs or peptide-drug conjugates have been widely explored in new drug development or drug release. Previously, peptide drugs were seen as merely mimicking hormones or endogenous peptides in the body, but they are now being reborn to be engineered into new therapies. In the past, few clinical patients used peptide drugs for antitumor therapy. However, as the limitations of peptide drugs are solved, the strong anti-tumor effect of peptide drugs has attracted the attention of researchers and the pipeline layout of more and more companies. Various therapeutic strategies combining cytotoxic antitumor drugs and functional peptides have been developed, and their effects are expected to be better than small molecule drugs, antibodies, and ADCs.